Potassium
Potassium is a chemical element in the periodic table. It has the symbol K. This symbol is taken from the Latin word kalium. Potassium's atomic number is 19. It has 19 protons and electrons. Potassium is not found as an element in nature, because it is so reactive.
 Potassium pearls (in paraffin oil, ~5 mm each) | ||||||||||||||||||||||||||||
| General properties | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pronunciation | /pəˈtæsiəm/ | |||||||||||||||||||||||||||
| Appearance | silvery gray | |||||||||||||||||||||||||||
| Standard atomic weight (Ar, standard) | 39.0983(1)[1] | |||||||||||||||||||||||||||
| Potassium in the periodic table | ||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||
| Atomic number (Z) | 19 | |||||||||||||||||||||||||||
| Group | group 1 (alkali metals) | |||||||||||||||||||||||||||
| Period | period 4 | |||||||||||||||||||||||||||
| Block | s-block | |||||||||||||||||||||||||||
| Element category | alkali metal | |||||||||||||||||||||||||||
| Electron configuration | [Ar] 4s1 | |||||||||||||||||||||||||||
Electrons per shell | 2, 8, 8, 1 | |||||||||||||||||||||||||||
| Physical properties | ||||||||||||||||||||||||||||
| Phase at STP | K: Solid | |||||||||||||||||||||||||||
| Melting point | 336.7 K (63.5 °C, 146.3 °F) | |||||||||||||||||||||||||||
| Boiling point | 1032 K (759 °C, 1398 °F) | |||||||||||||||||||||||||||
| Density (near r.t.) | 0.862 g/cm3 | |||||||||||||||||||||||||||
| when liquid (at m.p.) | 0.828 g/cm3 | |||||||||||||||||||||||||||
| Critical point | 2223 K, 16 MPa[2] | |||||||||||||||||||||||||||
| Heat of fusion | 2.33 kJ/mol | |||||||||||||||||||||||||||
| Heat of vaporization | 76.9 kJ/mol | |||||||||||||||||||||||||||
| Molar heat capacity | 29.6 J/(mol·K) | |||||||||||||||||||||||||||
| Atomic properties | ||||||||||||||||||||||||||||
| Oxidation states | −1, +1 (a strongly basic oxide) | |||||||||||||||||||||||||||
| Electronegativity | Pauling scale: 0.82 | |||||||||||||||||||||||||||
| Ionization energies |
| |||||||||||||||||||||||||||
| Atomic radius | empirical: 227 pm | |||||||||||||||||||||||||||
| Covalent radius | 203±12 pm | |||||||||||||||||||||||||||
| Van der Waals radius | 275 pm | |||||||||||||||||||||||||||
| Spectral lines of potassium | ||||||||||||||||||||||||||||
| Other properties | ||||||||||||||||||||||||||||
| Natural occurrence | K: Primordial | |||||||||||||||||||||||||||
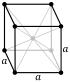
| Crystal structure | body-centered cubic (bcc) | |||||||||||||||||||||||||||
| Speed of sound thin rod | 2000 m/s (at 20 °C) | |||||||||||||||||||||||||||
| Thermal expansion | 83.3 µm/(m·K) (at 25 °C) | |||||||||||||||||||||||||||
| Thermal conductivity | 102.5 W/(m·K) | |||||||||||||||||||||||||||
| Electrical resistivity | 72 nΩ·m (at 20 °C) | |||||||||||||||||||||||||||
| Magnetic ordering | paramagnetic[3] | |||||||||||||||||||||||||||
| Magnetic susceptibility | +20.8·10−6 cm3/mol (298 K)[4] | |||||||||||||||||||||||||||
| Young's modulus | 3.53 GPa | |||||||||||||||||||||||||||
| Shear modulus | 1.3 GPa | |||||||||||||||||||||||||||
| Bulk modulus | 3.1 GPa | |||||||||||||||||||||||||||
| Mohs hardness | 0.4 | |||||||||||||||||||||||||||
| Brinell hardness | 0.363 MPa | |||||||||||||||||||||||||||
| CAS Number | 7440-09-7 | |||||||||||||||||||||||||||
| History | ||||||||||||||||||||||||||||
| Discovery and first isolation | Humphry Davy (1807) | |||||||||||||||||||||||||||
| Main isotopes of potassium | ||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||
Potassium has two stable isotopes, with 20 or 22 neutrons. Its atomic mass is 39.098. The unstable isotope with 21 neutrons is one of the most common radioactive materials.
Properties
Physical properties
Potassium is a soft gray metal. It can be cut easily with a knife. Its melting point is 63 degrees Celsius (145.4 degrees Fahrenheit). It melts at a very low temperature. It is an alkali metal. It is the second lightest metal, after lithium.
Chemical properties
Potassium reacts in many chemical reactions similar to sodium and other alkali metals. It tarnishes in air to produce a whitish oxidized layer on the surface. This is why it is stored in oil. It also reacts very fast with water, which is another reason for its storage in oil. The hydrogen produced during its reaction with water can burst into flames when a large amount of potassium is added to water. Potassium hydroxide is also produced. Potassium also burns in air easily, to make the peroxide or the superoxide.
Chemical compounds
Potassium compounds are only in one oxidation state: +1. Potassium ions are colorless and similar to sodium ions. Potassium chloride can be used as a substitute for table salt. Potassium hydroxide is used in the electrolyte of alkaline cells. Most potassium compounds are nontoxic. If they are toxic, it is because of the anion. Potassium chromate is colored because of the chromate, not the potassium. Potassium chromate is toxic because of the chromate, not the potassium.
- Chrome alum
- Potash alum
- Potassium arsenate, oxidizing agent, toxic
- Potassium arsenite, colorless solid, toxic
- Potassium bromate, colorless, oxidizing agent, used in flour
- Potassium bromide, colorless, used as sedative
- Potassium carbonate, colorless, reacts with acids
- Potassium chlorate, used in matches and explosives
- Potassium chloride, salt substitute
- Potassium chromate, yellow solid, oxidizing agent, toxic
- Potassium dichromate, red solid, oxidizing agent, toxic
- Potassium fluoride, used to make fluorine, corrosive
- Potassium hydroxide, also known as potash, caustic, strong base, white solid
- Potassium hypomanganate, bright blue hypomanganate, rare
- Potassium iodate, used to supply iodine
- Potassium iodide, used to supply iodine
- Potassium manganate, used to make potassium permanganate
- Potassium nitrate, used in gunpowder
- Potassium perchlorate, used in some rockets
- Potassium periodate
- Potassium permanganate, purple, disinfectant, oxidizing agent
- Potassium sulfide, reacts with water
- Potassium sulfite, used in food preservation
- Potassium sulfate, colorless, used in fertilizers
Name
The word potassium comes from the word "potash". Potash is a mixture of potassium carbonate and potassium hydroxide that has been used for a very long time. In past centuries potash was made from ashes in pots. It is used to make fertilizer, soap, and glass.
Occurrence
Potassium does not occur in nature because it is too reactive. It is found in minerals, though. It is extracted from them by electrolysis of potassium hydroxide or potassium chloride. The potassium hydroxide or potassium chloride has to be melted at a very high temperature.
Uses
Potassium metal is used to absorb water from solvents. It is also used in some scientific instruments.
Potassium compounds are used in soap, fertilizer, explosives, and matches.
Nutrition
Potassium ions are very important to organisms. That is why fertilizers have potassium compounds in them. The ions send messages from cells to other cells. It helps biological membranes depolarize. This means go from a negative to a positive electrical charge. This is needed for muscles to contract (get shorter and move things.) It is needed for the heart to beat (push blood through blood vessels.) If the potassium level in the blood is too high or too low it can cause death because the heart stops.[6] A few good sources of potassium are bananas, apricots and raisins.
Safety
Potassium metal is very dangerous and can form an explosive coating if it is kept in air. It also reacts violently with water, spewing corrosive liquid. Potassium compounds are not normally dangerous, unless they contain a toxic anion like chromate or chlorate.
PotassiumChemical Compounds Media
The flame test of potassium.
Sylvite from New Mexico
Monte Kali, a potash mining and beneficiation waste heap in Hesse, Germany, consisting mostly of sodium chloride.
Related pages
Other websites
- Potassium -Citizendium
References
- ↑ Meija, J.; Coplen, T. B.; Berglund, M.; Brand, W.A.; De Bièvre, P.; Gröning, M.; Holden, N.E.; Irrgeher, J.; Loss, R.D.; Walczyk, T.; Prohaska, T. (2016). "Atomic weights of the elements 2013 (IUPAC Technical Report)". Pure and Applied Chemistry. 88 (3): 265–91. doi:10.1515/pac-2015-0305.
{{cite journal}}: Unknown parameter|displayauthors=ignored (|display-authors=suggested) (help) - ↑ Haynes, William M., ed. (2011). CRC Handbook of Chemistry and Physics (92nd ed.). Boca Raton, FL: CRC Press. p. 4.122. ISBN 1439855110.
- ↑ Magnetic susceptibility of the elements and inorganic compounds, in Lide, D. R., ed. (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5.
- ↑ Weast, Robert (1984). CRC, Handbook of Chemistry and Physics. Boca Raton, Florida: Chemical Rubber Company Publishing. pp. E110. ISBN 0-8493-0464-4.
- ↑ "Potassium". Retrieved 13 October 2010.[dead link]
- ↑ Whelton, P.; He, J.; Cutler, J.; Brancati, F.; Appel, L.; Follmann, D.; Klag, M. (1997). "Effects of oral potassium on blood pressure. Meta-analysis of randomized controlled clinical trials". JAMA. 277 (20): 1624–1632. doi:10.1001/JAMA.1997.03540440058033. PMID 9168293. S2CID 25937399.