Electron cloud
Electron cloud is an informal way to describe an atomic orbital.
The electron cloud is not really a thing. An electron cloud model is different from the older Bohr atomic model by Niels Bohr. Bohr talked about electrons orbiting the nucleus. Explaining the behavior of these electron "orbits" was a key issue in the development of quantum mechanics.[1]
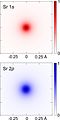
The electron cloud model says that we cannot know exactly where an electron is at any given time, but the electrons are more likely to be in specific areas. These areas are specified by orbitals. The orbitals are specified by shells and sub-orbitals. In the Bohr model, electrons are assigned to different shells. The shells, k,l,m,n,o,p,q, each represent different levels of energy, and are also called energy levels. The sub-orbitals; s,p,d,f, are regions where it will be more likely to find electrons, and can each hold a different number of electrons. the s,p,d,f orbitals are all shaped differently. This can be proven by the repeating patterns of chemical properties in the periodic table. Using quantum mechanics, chemists can use the electron cloud model to assign electrons to different atomic orbitals. Atomic orbitals also explain the patterns in the periodic table.
The electron cloud model was developed in 1926 by Erwin Schrödinger and Werner Heisenberg. The model is a way to help visualize the most probable position of electrons in an atom. The electron cloud model is currently the accepted model of an atom. The electrons were traveling at such a high speed that there was no predictable time or place for the electron to be. The electrons were like a blur which is where they got the 'Electron Cloud' from.
According to Bohr's calculations for a hydrogen atom, the electron under normal conditions always stays at a certain distance from the nucleus. This distance is called the Bohr radius and is approximately 0.529 Å (0.529×10−10 m). But according to the wave mechanical or cloud concept model, the electron keeps on moving away or towards the nucleus and the maximum probability of locating it lies at a distance of 0.529 Å from the nucleus. In other words, the radius of the electron cloud or the radius of maximum probability is 0.529 Å.
Electron Cloud Media
3D views of some hydrogen-like atomic orbitals showing probability density and phase (g orbitals and higher not shown)
The Rutherford–Bohr model of the hydrogen atom
Atomic orbitals and periodic table construction
References
- ↑ Bryson, Bill 2003 (2004). A short history of nearly everything. Broadway Books. pp. 141–143. ISBN 0-7679-0818-X.
+{{{1}}}−{{{2}}}