Template:Infobox unbinilium
- Temp: comparing dataset and input-parameter here to check. No effect in article. [January 2019]
- DIFF
- (parameter is empty) -- infobox
- [Og] 8s2 (predicted)[1] -- dataset
- Using symb-to-elconfig dataset now. DIFFs marked here may be caused by technical issues wrt reference handling, not actual diffs. -DePiep (talk) 20:17, 3 February 2019 (UTC)
| General properties | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Pronunciation | /ˌuːnbaɪˈnɪliəm/ | ||||||||||
| Alternative names | element 120, eka-radium | ||||||||||
| Mass number | unconfirmed: 299 (most stable isotope) | ||||||||||
| Unbinilium in the periodic table | |||||||||||
| |||||||||||
| Atomic number (Z) | 120 | ||||||||||
| Group | group 2 (alkaline earth metals) | ||||||||||
| Period | period 8 | ||||||||||
| Block | s-block | ||||||||||
| Element category | alkaline earth metal (predicted) | ||||||||||
| Electron configuration | [Og] 8s2 (predicted)[1] | ||||||||||
Electrons per shell | 2, 8, 18, 32, 32, 18, 8, 2 (predicted) | ||||||||||
| Physical properties | |||||||||||
| Phase at STP | Ubn: Unknown phase (predicted)[1][2] | ||||||||||
| Melting point | 953 K (680 °C, 1256 °F) (predicted)[1] | ||||||||||
| Boiling point | 1973 K (1700 °C, 3092 °F) (predicted)[3] | ||||||||||
| Density (near r.t.) | 7 g/cm3 (predicted)[1] | ||||||||||
| Heat of fusion | 8.03–8.58 kJ/mol (extrapolated)[2] | ||||||||||
| Atomic properties | |||||||||||
| Oxidation states | (+1),[4] (+2), (+4) (predicted)[1] | ||||||||||
| Electronegativity | Pauling scale: 0.91 (predicted)[5] | ||||||||||
| Ionization energies | |||||||||||
| Atomic radius | empirical: 200 pm (predicted)[1] | ||||||||||
| Covalent radius | 206–210 pm (extrapolated)[2] | ||||||||||
| Other properties | |||||||||||
| Natural occurrence | Ubn: | ||||||||||
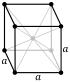
| Crystal structure | body-centered cubic (bcc) (extrapolated)[7] | ||||||||||
| CAS Number | 54143-58-7 | ||||||||||
| History | |||||||||||
| Naming | IUPAC systematic element name | ||||||||||
| Main isotopes of unbinilium | |||||||||||
| |||||||||||
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Hoffman, Darleane C.; Lee, Diana M.; Pershina, Valeria (2006). "Transactinides and the future elements". In Morss; Edelstein, Norman M.; Fuger, Jean (eds.). The Chemistry of the Actinide and Transactinide Elements (3rd ed.). Dordrecht, The Netherlands: Springer Science+Business Media. ISBN 1-4020-3555-1.
- ↑ 2.0 2.1 2.2 2.3 Bonchev, Danail; Kamenska, Verginia (1981). "Predicting the properties of the 113-120 transactinide elements". Journal of Physical Chemistry. American Chemical Society. 85 (9): 1177–1186. doi:10.1021/j150609a021.
- ↑ Fricke, B.; Waber, J. T. (1971). "Theoretical Predictions of the Chemistry of Superheavy Elements" (PDF). Actinides Reviews. 1: 433–485. Retrieved 7 August 2013.
- ↑ Thayer, John S. (2010). "Relativistic Effects and the Chemistry of the Heavier Main Group Elements". Relativistic Methods for Chemists: 84. doi:10.1007/978-1-4020-9975-5_2.
- ↑ Pershina, V.; Borschevsky, A.; Anton, J. (2012). "Theoretical predictions of properties of group-2 elements including element 120 andtheir adsorption on noble metal surfaces". The Journal of Chemical Physics. 136 (134317). doi:10.1063/1.3699232. This article gives the Mulliken electronegativity as 2.862, which has been converted to the Pauling scale via χP = 1.35χM1/2 − 1.37.
- ↑ Pershina, Valeria. "Theoretical Chemistry of the Heaviest Elements". In Schädel, Matthias; Shaughnessy, Dawn (eds.). The Chemistry of Superheavy Elements (2nd ed.). Springer Science & Business Media. p. 154. ISBN 9783642374661.
- ↑ Seaborg, Glenn T. (1969). "Prospects for further considerable extension of the periodic table" (PDF). Journal of Chemical Education. 46 (10): 626–634. doi:10.1021/ed046p626. Retrieved 22 February 2018.
- ↑ (2016) "Remarks on the Fission Barriers of SHN and Search for Element 120" in Exotic Nuclei. : 155–164.